单克隆抗体dankelongkangti
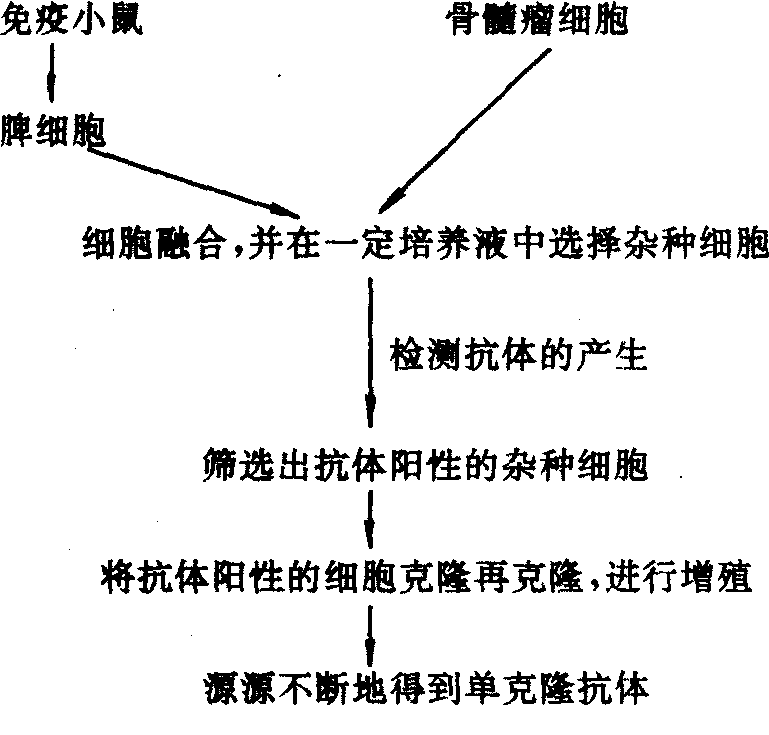
是单个细胞增殖形成的细胞群所产生的抗体,因它是针对单一抗原决定簇的,所以具有很强的专一性。获得单克隆抗体的方法,主要利用细胞融合技术,将正常产生抗体的脾B细胞和不产生抗体的骨髓瘤细胞融合,融合细胞既保持着骨髓瘤细胞系的迅速生长和传代的性能,又具有B细胞产生特异性抗体的特性,所以能在体外连续培养。单克隆抗体就是筛选出来的单个融合细胞(杂交瘤细胞)的产物。产生单克隆抗体的大致过程如下:
由于单克隆抗体有较高的灵敏度和特异性,所以有很大的应用价值,可进行一些疾病的诊断和治疗,如对绒毛膜促性腺激素、生长激素等的测定,以诊断有关疾病,以及进行对乙型肝炎及某些肿瘤的诊断等,它和药物结合能制成“导弹药物”到达体内专一部位,以进行对某些疾病的治疗。在科研上,单克隆抗体也是一种有力的研究工具。
单克隆抗体monoclonal antibody
由克隆化的B细胞杂交瘤所产生的、结构高度均质的,针对特定抗原的免疫球蛋白。1975年克勒 (G. Kohler)和米尔斯坦(C.Milstein) 建立淋巴细胞杂交瘤技术,他们将具有代谢缺陷型的小鼠骨髓瘤细胞与经抗原免疫后的小鼠脾细胞,在聚乙二醇的作用下融合,然后在含次黄嘌呤(H)、氨基蝶呤(A)和胸腺嘧啶核苷 (T)的HAT培养基上选择培养。骨髓瘤细胞因缺乏利用次黄嘌呤的酶,不能生长,未融合的脾细胞在体外培养时间一般不超过两周,只有融合的瘤细胞能在 HAT培养基上生长。通过抗体检测和细胞的克隆化,即可获得克隆化的杂交瘤细胞株。此杂交瘤细胞具有两个亲代细胞的遗传性,它既能无限增殖,又能分泌特定的抗体。由此杂交瘤细胞株所产生的抗体即单克隆抗体。杂交瘤细胞株可在液氮中长期冻存,用时解冻复苏,即可在细胞培养上或接种到同系小鼠腹腔中进行生产。杂交瘤细胞株一旦选育成功,只要其染色体不丢失,就可长期用来生产针对特定抗原的单克隆抗体。
常规免疫血清中包含有针对不同抗原决定簇的抗体和多种免疫球蛋白类型,具有高度异质性,它由多个B细胞克隆所产生,故称为多克隆抗体。与之相比,单克隆抗体由单一B细胞克隆所产生,只针对一种抗原决定簇,属于一种免疫球蛋白类型,具有高度均质性。用一种抗原免疫小鼠,可以筛选出许多株针对不同抗原决定簇和属于不同免疫球蛋白类型的单克隆抗体,它们具有不同免疫生物学活性。因此,可根据试验目的选择所需的克隆株。只要筛选出针对性强的单克隆抗体,就很容易区别两种近似物质之间的差异。如不同菌株、毒株的鉴别,淋巴细胞亚群和组织相容抗原分类; 病毒和细菌的保护性抗原分析等。这些都是应用多克隆抗体所难以做到的。
由于单克隆抗体特异性强、效价高、均质性好,用以制备标准化诊断试剂,可大大提高免疫诊断水平。除鉴别诊断外,对各类活性物质的分析,各种细胞产物及其受体研究和遗传工程中基因表达产物的检测等都将发挥巨大的作用。此外,在免疫防治中,也具有广泛的应用前景。如将蓖麻毒素或白喉毒素等连接于针对肿瘤特异性抗原的单克隆抗体上,制成免疫导向药物,可定向地杀伤肿瘤细胞。此类制剂称为免疫毒素,已开始应用于肿瘤治疗。中和性单克隆抗体不仅可用于病毒病的防治,还可用以制备抗独特型抗体疫苗,供主动免疫之用。
单克隆抗体
简称“单抗”。通过单克隆的培养而产生的一种结构、氨基酸排列顺序和特异性完全一致的抗体。具有高度的特异性和专一性,只与一种抗原决定簇起反应,广泛用于医学和生物学各门学科的研究和临床疾病的诊断及治疗。单抗的问世,被认为是医学和生物学领域的一场革命。由于单抗可携带放射性同位素、药物、毒素等到达机体内部的特定病变部位(如肿瘤),从而起到诊断和治疗的作用,因此又被称为“生物导弹”。但单抗的这种用途还处于临床实验阶段。(参见“B细胞杂交瘤”)
单克隆抗体
杂交瘤细胞在培养过程中产生的单一类型的抗体。用于疾病的诊断、流行病监测、疫苗研制、以及基础生物学和医学方面的研究。
单克隆抗体monoclonal antibody
由克隆化的B细胞杂交瘤所产生、结构高度均质、针对特定抗原的免疫球蛋白。1975年克勒(G.Kohler)和米尔斯坦(C.Milstein)建立淋巴细胞杂交瘤技术,他们将具有代谢缺陷型的小鼠骨髓瘤细胞与经抗原免疫后的小鼠脾细胞,在聚乙二醇的作用下进行融合,再在含次黄嘌呤(H)、氨基蝶呤(A)和胸腺嘧啶核苷(T)的HAT培养基上选择培养。骨髓瘤细胞因缺少利用次黄嘌呤的酶,不能生长,未经融合的脾细胞在体外培养时间一般不超过两周,只有融合的瘤细胞能在HAT培养基上生长。通过抗体检测和细胞的克隆化,可获得克隆化的杂交瘤细胞株。杂交瘤细胞具有双亲细胞的遗传性,既能无限增殖,又能分泌特定的抗体,这种杂交瘤细胞株所产生的抗体即单克隆抗体。单克隆抗体特异性强、效价高、均质性好,可制备标准化诊断试剂,大大提高免疫诊断水平;对各类活性物质分析、各种细胞产物及其受体研究和遗传工程中基因表达产物的检测中都能发挥巨大作用;另在免疫防治中也具有广泛的应用前景。