- Experiment design
- Appropriateness
- Model
- Estimate of variance
- Advantages
- Disadvantages
- Ongoing work
- References
}}
A stepped-wedge trial (or SWT) is a type of randomised controlled trial (or RCT), a scientific experiment which is structured to reduce bias when testing new medical treatments, social interventions, or other testable hypotheses. In a traditional RCT, a part of the participants in the experiment are simultaneously and randomly assigned to a group that receives the treatment (the "treatment group") and another part to a group that does not (the "control group"). In an SWT, typically a logistical constraint prevents the simultaneous treatment of some participants, and instead, all or most participants receive the treatment in waves or "steps".
For instance, suppose a researcher wanted to measure whether teaching college students how to make several meals increased their propensity to cook at home instead of eating out. In a traditional RCT, a sample of students would be selected and some would be trained on how to cook these meals, whereas the some others would not. Both groups would be monitored to see how frequently they ate out. In the end, the number of times the treatment group ate out would be compared to the number of times the control group ate out, most likely with a t-test or some variant. If, however, the researcher could only train a limited number of students each week, then the researcher could employ an SWT, randomly assigning students to which week they would be trained.
The term "stepped wedge" was coined by the Gambia Hepatitis Intervention Study due to the stepped-wedge shape that is apparent from a schematic illustration of the design.[1] The crossover is in one direction, typically from control to intervention, with the intervention not removed once implemented. The stepped-wedge design can be used for individually randomized trials,[2][3] i.e., trials where each individual is treated sequentially, but is more commonly used as a cluster randomized trial (CRT).[4]
Experiment design
The stepped-wedge design involves the collection of observations during a baseline period in which no clusters are exposed to the intervention. Following this, at regular intervals, or steps, a cluster (or group of clusters) is randomized to receive the intervention[4][5] and all participants are once again measured.[6] This process continues until all clusters have received the intervention. Finally, one more measurement is made after all clusters have received the intervention.
Appropriateness
Hargreaves and colleagues offer a series of five questions that researchers should answer to decide whether SWT is indeed the optimal design, and how to proceed in every step of the study.[7] Specifically, researchers should be able to identify:
- The reasons SWT is the preferred design
- If the measuring a treatment effect is the primary goal of research, SWT may not be the optimal design. SWTs are appropriate when the research focus is on the effectiveness of the treatment rather than on its mere existence. Overall, if the study is pragmatic (i.e. seeks primarily to implement a certain policy) logistical and other practical concerns are considered to be the best reasons to turn to a stepped wedge design. On the contrary, if the study is explanatory (i.e. seeks to study the cause of an effect), the benefits are significant but so are the challenges. Repeated interventions and the corollary training workload of interviewers over time, minimizing attrition, and ensuring compliance and ignorability can increase costs, and undermine unbiasedness and efficiency. Moreover, dealing with ethical issues related to postponing the intervention for some clusters is also crucial.
- Which SWT design is more suitable
- SWTs can feature three main designs employing a closed cohort, an open cohort, and a continuous recruitment with short exposure.[8] Typically, in the first design all subjects participate from the beginning of the experiment and until its completion, and the outcomes are measured repeatedly at fixed time points which may or may not be related to each step. In the open cohort design, outcomes are measured similarly to the former design, but new subjects can enter the study, and some participants from an early stage can leave before the completion. Only a part of the subjects are exposed from the start, and more are gradually exposed in subsequent steps. Thus, the time of exposure varies for each subject. In continuous recruitment with short exposure , very few or no subjects participate in the beginning of the experiment but more become eligible, and are exposed to short intervention gradually. In this design, each subject is assigned to either the treatment or the control condition. Since participants are assigned to either the treatment or the control group, the risk of carry-over effects, which may be a challenge for closed and open cohort designs, is minimal.
- Which analysis strategy is appropriate
- Linear Mixed Models (LMM), Generalized Linear Mixed Models (GLMM), and Generalized Estimating Equations (GEE) are the principal estimators that are recommended for analyzing the results. While LMM offers higher power than GLMM and GEE, it can be inefficient if the size of clusters vary, and the response is not continuous and normally distributed. If any of those assumptions is violated, GLMM and GEE are preferred.
- How big the sample should be
- Power analysis and sample size calculation are available. Generally, SWTs require smaller sample size to detect effects since they leverage both between and within-cluster comparisons.[10][9]
- Best practices for reporting the design and results of the trial
- Reporting the design, sample profile, and results can be challenging, since no Consolidated Standards Of Reporting Trials (CONSORT) have been designated for SWTs. However, some studies have provided both formalizations and flow charts that help reporting results, and sustaining a balanced sample across the waves.[10]
Model
While there are several other potential methods for modeling outcomes in an SWT,[11] the work of Hussey and Hughes[6] "first described methods to determine statistical power available when using a stepped wedge design."[11] What follows is their design.
Suppose there are  samples divided into
samples divided into  clusters. At each time point
clusters. At each time point  , preferably equally spaced in actual time, some number of clusters are treated. Let
, preferably equally spaced in actual time, some number of clusters are treated. Let  be
be  if cluster
if cluster  has been treated at time
has been treated at time  and
and  otherwise. In particular, note that if
otherwise. In particular, note that if  then
then  .
.
For each participant 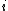 in cluster
in cluster  , measure the outcome to be studied
, measure the outcome to be studied  at time
at time  . Note that the notation allows for clustering by including
. Note that the notation allows for clustering by including  in the subscript of
in the subscript of  ,
,  ,
,  , and
, and 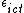 . We model these outcomes as:
. We model these outcomes as: 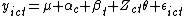 where:
where:
-
 is a grand mean,
is a grand mean, -
 is a random, cluster-level effect on the outcome,
is a random, cluster-level effect on the outcome, -
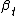 is a time point-specific fixed effect,
is a time point-specific fixed effect, -
 is the measured effect of the treatment, and
is the measured effect of the treatment, and -
 is the residual noise.
is the residual noise.
This model can be viewed as a Hierarchical linear model where at the lowest level 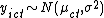 where
where 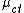 is the mean of a given cluster at a given time, and at the cluster level, each cluster mean
is the mean of a given cluster at a given time, and at the cluster level, each cluster mean 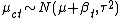 .
.
Estimate of variance
The design effect (estimate of unit variance) of a stepped wedge design is given by the formula:[10]
 where:
where:
- ρ is the intra-cluster correlation (ICC),
- n is the number of subjects within a cluster (which is assumed to be constant),
- k is the number of steps,
- t is the number of measurements after each step, and
- b is the number of baseline measurements.
To calculate the sample size it is needed to apply the simple formula:[10]

where:
- Nsw is the required sample size for the SWT
- Nu is the total unadjusted sample size that would be required for a traditional RCT.
Note that increasing either k, t, or b will result to decreasing the required sample size for an SWT.
Further, the required cluster c size is given by:[10]

To calculate how many clusters cs need to switch from the control to the treatment condition, the following formula is available:[10]

If c and cs are not integers they need to be rounded to the next larger integer, and distributed as evenly as possible among k.
Advantages
Stepped wedge design features many comparative advantages to traditional RCTs. First, SWTs are most appropriate both ethically and practically when the intervention is expected to produce a positive outcome. Since all subjects will eventually receive the benefits of the intervention, ethical concerns can be appeased, and the recruitment of participants may become easier.[10] Secondly, SWTs "can reconcile the need for robust evaluations with political or logistical constraints."[11] Specifically, it can be used to measure the effects of treatment when resources for performing an intervention are scarce.
Thirdly, since each cluster receives both the control and the treatment condition by the end of the trial, both between and within-cluster comparisons are possible. This way statistical power increases while keeping the sample significantly smaller than it would be needed in a traditional RCT.[10] Finally, because each cluster switches randomly from control to treatment condition in different time points, it is possible to examine time effects.[10] For example, it is possible to study how repeated or long-term exposure to experimental stimuli affects the efficiency of the treatment. Repeated measurements in regular time frames can average the noise out, which in turn increases the precision of estimates. This advantage becomes most apparent when measurement is noisy, and outcome autocorrelation is low.[12]
Disadvantages
SWT may suffer from certain drawbacks. First, since in SWTs the study period lasts longer and all the subjects eventually receive the treatment, costs may increase significantly.[10] Because the design can be expensive, SWTs may not be the optimal solution when measurement precision and outcome autocorrelation are high.[12] Moreover, since everyone is eventually treated, SWTs do not facilitate downstream analysis.
Secondly, in an SWT, more clusters are exposed to the intervention at later than earlier time periods. As such, it is possible that an underlying temporal trend may confound the intervention effect, and so the confounding effect of time must be accounted for in both pre-trial power calculations and post-trial analysis.[4][13][11] Specifically, in post-trial analysis, the use of generalized linear mixed models or generalized estimating equations is recommended.[10]
Finally, the design and analysis of stepped-wedge trials is therefore more complex than for other types of randomized trials. Previous systematic reviews highlighted the poor reporting of sample size calculations and a lack of consistency in the analysis of such trials.[4][5] Hussey and Hughes were the first authors to suggest a structure and formula for estimating power in stepped-wedge studies in which data was collected at each and every step.[6] This has now been expanded for designs in which observations are not made at each step as well as multiple layers of clustering.[14] Additionally, a design effect (used to inflate the sample size of an individually randomized trial to that required in a cluster trial) has been established,[15] which has shown that the stepped wedge CRT could reduce the number of patients required in the trial compared to other designs.[15][16]
Ongoing work
The number of studies using the design have been on the increase. In 2015, a thematic series was published in the journal Trials.[17] In 2016, the first international conference dedicated to the topic was held at the University of York.[18][19]
References
2. ^{{cite journal | vauthors = Ratanawongsa N, Handley MA, Quan J, Sarkar U, Pfeifer K, Soria C, Schillinger D | title = Quasi-experimental trial of diabetes Self-Management Automated and Real-Time Telephonic Support (SMARTSteps) in a Medicaid managed care plan: study protocol | journal = BMC Health Services Research | volume = 12 | issue = | pages = 22 | date = January 2012 | pmid = 22280514 | pmc = 3276419 | doi = 10.1186/1472-6963-12-22 }}
3. ^{{cite journal | vauthors = Løhaugen GC, Beneventi H, Andersen GL, Sundberg C, Østgård HF, Bakkan E, Walther G, Vik T, Skranes J | title = Do children with cerebral palsy benefit from computerized working memory training? Study protocol for a randomized controlled trial | journal = Trials | volume = 15 | issue = | pages = 269 | date = July 2014 | pmid = 24998242 | pmc = 4226979 | doi = 10.1186/1745-6215-15-269 }}
4. ^1 2 3 {{cite journal | vauthors = Brown CA, Lilford RJ | title = The stepped wedge trial design: a systematic review | journal = BMC Medical Research Methodology | volume = 6 | issue = | pages = 54 | date = November 2006 | pmid = 17092344 | pmc = 1636652 | doi = 10.1186/1471-2288-6-54 }}
5. ^1 {{cite journal | vauthors = Mdege ND, Man MS, Taylor Nee Brown CA, Torgerson DJ | title = Systematic review of stepped wedge cluster randomized trials shows that design is particularly used to evaluate interventions during routine implementation | journal = Journal of Clinical Epidemiology | volume = 64 | issue = 9 | pages = 936–48 | date = September 2011 | pmid = 21411284 | doi = 10.1016/j.jclinepi.2010.12.003 }}
6. ^1 2 {{cite journal | vauthors = Hussey MA, Hughes JP | title = Design and analysis of stepped wedge cluster randomized trials | journal = Contemporary Clinical Trials | volume = 28 | issue = 2 | pages = 182–91 | date = February 2007 | pmid = 16829207 | doi = 10.1016/j.cct.2006.05.007 }}
7. ^{{cite journal | vauthors = Hargreaves JR, Copas AJ, Beard E, Osrin D, Lewis JJ, Davey C, Thompson JA, Baio G, Fielding KL, Prost A | title = Five questions to consider before conducting a stepped wedge trial | language = En | journal = Trials | volume = 16 | issue = 1 | pages = 350 | date = August 2015 | pmid = 26279013 | pmc = 4538743 | doi = 10.1186/s13063-015-0841-8 }}
8. ^{{cite journal | vauthors = Copas AJ, Lewis JJ, Thompson JA, Davey C, Baio G, Hargreaves JR | title = Designing a stepped wedge trial: three main designs, carry-over effects and randomisation approaches | language = En | journal = Trials | volume = 16 | issue = 1 | pages = 352 | date = August 2015 | pmid = 26279154 | pmc = 4538756 | doi = 10.1186/s13063-015-0842-7 }}
9. ^{{cite journal | vauthors = Baio G, Copas A, Ambler G, Hargreaves J, Beard E, Omar RZ | title = Sample size calculation for a stepped wedge trial | language = En | journal = Trials | volume = 16 | issue = 1 | pages = 354 | date = August 2015 | pmid = 26282553 | pmc = 4538764 | doi = 10.1186/s13063-015-0840-9 }}
10. ^{{cite journal | vauthors = Gruber JS, Reygadas F, Arnold BF, Ray I, Nelson K, Colford JM | title = A stepped wedge, cluster-randomized trial of a household UV-disinfection and safe storage drinking water intervention in rural Baja California Sur, Mexico | journal = The American Journal of Tropical Medicine and Hygiene | volume = 89 | issue = 2 | pages = 238–45 | date = August 2013 | pmid = 23732255 | pmc = 3741243 | doi = 10.4269/ajtmh.13-0017 }}
11. ^1 2 3 {{cite journal | vauthors = Hemming K, Haines TP, Chilton PJ, Girling AJ, Lilford RJ | title = The stepped wedge cluster randomised trial: rationale, design, analysis, and reporting | journal = BMJ | volume = 350 | pages = h391 | date = February 2015 | pmid = 25662947 | doi = 10.1136/bmj.h391 }}
12. ^1 {{cite journal | vauthors = McKenzie D |date=November 2012|title=Beyond baseline and follow-up: The case for more T in experiments Author links open overlay panel |journal=Journal of Development Economics |volume = 99 | issue = 2 | pages = 210–221 }}
13. ^{{cite journal | vauthors = Van den Heuvel ER, Zwanenburg RJ, Van Ravenswaaij-Arts CM | title = A stepped wedge design for testing an effect of intranasal insulin on cognitive development of children with Phelan-McDermid syndrome: A comparison of different designs | journal = Statistical Methods in Medical Research | volume = 26 | issue = 2 | pages = 766–775 | date = April 2017 | pmid = 25411323 | doi = 10.1177/0962280214558864 }}
14. ^{{cite journal | vauthors = Hemming K, Lilford R, Girling AJ | title = Stepped-wedge cluster randomised controlled trials: a generic framework including parallel and multiple-level designs | journal = Statistics in Medicine | volume = 34 | issue = 2 | pages = 181–96 | date = January 2015 | pmid = 25346484 | pmc = 4286109 | doi = 10.1002/sim.6325 }}
15. ^1 2 3 4 5 6 7 8 9 10 11 {{cite journal | vauthors = Woertman W, de Hoop E, Moerbeek M, Zuidema SU, Gerritsen DL, Teerenstra S | title = Stepped wedge designs could reduce the required sample size in cluster randomized trials | journal = Journal of Clinical Epidemiology | volume = 66 | issue = 7 | pages = 752–8 | date = July 2013 | pmid = 23523551 | doi = 10.1016/j.jclinepi.2013.01.009 }}
16. ^{{cite journal | vauthors = Keriel-Gascou M, Buchet-Poyau K, Rabilloud M, Duclos A, Colin C | title = A stepped wedge cluster randomized trial is preferable for assessing complex health interventions | journal = Journal of Clinical Epidemiology | volume = 67 | issue = 7 | pages = 831–3 | date = July 2014 | pmid = 24774471 | doi = 10.1016/j.jclinepi.2014.02.016 }}
17. ^{{cite journal|last1=Torgerson|first1=David | name-list-format = vanc |title=Stepped Wedge Randomized Controlled Trials|journal=Trials|date=2015|volume=16|page=350|url=http://www.biomedcentral.com/collections/SteppedWedge|access-date=17 February 2017}}
18. ^{{cite web | title = First International Conference on Stepped Wedge Trial Design |publisher =University of York |url= https://www.york.ac.uk/healthsciences/research/trials/sw_conf/ }}
19. ^{{cite journal |last1=Kanaan |first1=Mona |last2=Keding |first2=Ada |last3=Mdege |first3=Noreen |last4=Torgerson |first4=David | name-list-format = vanc |title=Proceedings of the First International Conference on Stepped Wedge Trial Design |journal= Trials |date=2016 |volume=17 | issue = Suppl 1 | pages = 311 | url = https://trialsjournal.biomedcentral.com/articles/supplements/volume-17-supplement-1 }}
- Aegyptosaurus baharijensis
- Aegyptus Province
- Aegyptus (Roman province)
- Aegyssus
- A. E. Harkavy
- A&E Home Video
- A. E. Hotchner
- A.E. Hotchner
- A. E. Houseman
- Aeife
- AEI Music
- A.E.I.O.U
- A.E.I.O.U. (album)
- AEIOU Encyclopedia
- AEIOU encylcopedia
- A-Eiropa
- AEJ
- A. E. Jacomb
- Aejang of Silla
- A.E.J. Collins
- A. E. J. Collins/scorecard
- A.E.J.Collins/scorecard
- AEJMC
- AEK Arena
- A.E.K. Arena