- Safe limit of partial pressure of oxygen
- Formula
- Tables of MOD
- See also
- Notes
- References
- Sources
In underwater diving activities such as saturation diving, technical diving and nitrox diving, the maximum operating depth (MOD) of a breathing gas is the depth below which the partial pressure of oxygen (pO2) of the gas mix exceeds an acceptable limit. This limit is based on risk of central nervous system oxygen toxicity, and is somewhat arbitrary, and varies depending on the diver training agency or Code of Practice, the level of underwater exertion planned and the planned duration of the dive, but is normally in the range of 1.2 to 1.6 bar.[1]
The MOD is significant when planning dives using gases such as heliox, nitrox and trimix because the proportion of oxygen in the mix determines a maximum depth for breathing that gas at an acceptable risk. There is a risk of acute oxygen toxicity if the MOD is exceeded.[1] The tables below show MODs for a selection of oxygen mixes. Atmospheric air contains approximately 21% oxygen, and has an MOD calculated by the same method.
Safe limit of partial pressure of oxygen
Acute, or central nervous system oxygen toxicity is a time variable response to the partial pressure exposure history of the diver and is both complex and not fully understood.
Central nervous system oxygen toxicity manifests as symptoms such as visual changes (especially tunnel vision), ringing in the ears (tinnitus), nausea, twitching (especially of the face), behavioural changes (irritability, anxiety, confusion), and dizziness. This may be followed by a tonic–clonic seizure consisting of two phases: intense muscle contraction occurs for several seconds (tonic phase); followed by rapid spasms of alternate muscle relaxation and contraction producing convulsive jerking (clonic phase). The seizure ends with a period of unconsciousness (the postictal state).{{sfn|Clark|Thom|2003|p=376}}{{sfn|U.S. Navy Diving Manual|2011|p=44|loc=vol. 1, ch. 3}} The onset of seizure depends upon the partial pressure of oxygen in the breathing gas and exposure duration. However, exposure time before onset is unpredictable, as tests have shown a wide variation, both amongst individuals, and in the same individual from day to day.{{sfn|Clark|Thom|2003|p=376}}{{sfn|U.S. Navy Diving Manual|2011|p=22|loc=vol. 4, ch. 18}}[3] In addition, many external factors, such as underwater immersion, exposure to cold, and exercise will decrease the time to onset of central nervous system symptoms.{{sfn|Donald, Part I|1947}} Decrease of tolerance is closely linked to retention of carbon dioxide.{{sfn|Lang|2001|p=82}}[4][5] Other factors, such as darkness and caffeine, increase tolerance in test animals, but these effects have not been proven in humans.[6][7]The maximum single exposure limits recommended in the NOAA Diving Manual are 45 minutes at 1.6 bar, 120 minutes at 1.5 bar, 150 minutes at 1.4 bar, 180 minutes at 1.3 bar and 210 minutes at 1.2 bar.[1]
Formula
The formula simply divides the absolute partial pressure of oxygen which can be tolerated (expressed in atmospheres or bar) by the fraction of oxygen in the breathing gas, to calculate the absolute pressure at which the mix can be breathed. (for example, 50% nitrox can be breathed at twice the pressure of 100% oxygen, so divide by 0.5, etc.). Of this total pressure which can be tolerated by the diver, 1 atmosphere is due to surface pressure of the Earth's air, and the rest is due to the depth in water. So the 1 atmosphere or bar contributed by the air is subtracted to give the pressure due to the depth of water. The pressure produced by depth in water, is converted to pressure in feet sea water (fsw) or metres sea water (msw) by multiplying with the appropriate conversion factor, 33 fsw per atmosphere, or 10 msw per bar.
In feet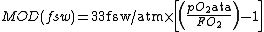
In which pO2 is the chosen maximum partial pressure of oxygen in atmospheres absolute and the FO2 is the fraction of oxygen in the mixture. For example, if a gas contains 36% oxygen (FO2 = 0.36) and the limiting maximum pO2 is chosen at 1.4 atmospheres absolute, the MOD in feet of seawater (fsw) {{refn|group=Notes|Feet sea water (fsw) is a unit of pressure. One fsw is equal to the hydrostatic pressure exerted by a standard sea water column of 1 foot height at normal Earth gravity. 33 fsw is approximately equal to one standard atmosphere (atm). A pressure indicated in fsw is gauge pressure (relative to surface pressure) unless specified.}} is 33 fsw/atm x [(1.4 ata / 0.36) - 1] = 95.3 fsw.[9]
In metres
In which pO2 is the chosen maximum partial pressure in oxygen in bar and the FO2 is the fraction of oxygen in the mixture. For example, if a gas contains 36% oxygen and the maximum pO2 is 1.4 bar, the MOD (msw) is 10 msw/bar x [(1.4 bar / 0.36) - 1] = 28.9 msw.
Tables of MOD
| MOD (fsw) | % oxygen | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 | 8 | 12 | 16 | 20 | 24 | 28 | 32 | 36 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | ||
| Maximum pO2 (bar) | 1.6 | 1287 | 627 | 407 | 297 | 231 | 187 | 156 | 132 | 114 | 99 | 73 | 55 | 42 | 33 | 26 | 20 |
| 1.5 | 1205 | 586 | 380 | 276 | 215 | 173 | 144 | 122 | 105 | 91 | 66 | 50 | 38 | 29 | 22 | 17 | |
| 1.4 | 1122 | 545 | 352 | 256 | 198 | 160 | 132 | 111 | 95 | 83 | 59 | 44 | 33 | 25 | 18 | 13 | |
| 1.3 | 1040 | 503 | 325 | 235 | 182 | 146 | 120 | 101 | 86 | 74 | 53 | 39 | 28 | 21 | 15 | 10 | |
| 1.2 | 957 | 462 | 297 | 215 | 165 | 132 | 108 | 91 | 77 | 66 | 46 | 33 | 24 | 17 | 11 | 7 | |
These depths are rounded down to the nearest foot.
| MOD (msw) | % oxygen | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 | 8 | 12 | 16 | 20 | 24 | 28 | 32 | 36 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | ||
| Maximum pO2 (bar) | 1.6 | 390 | 190 | 123 | 90 | 70 | 57 | 47 | 40 | 34 | 30 | 22 | 17 | 13 | 10 | 8 | 6 |
| 1.5 | 365 | 178 | 115 | 84 | 65 | 53 | 44 | 37 | 32 | 28 | 20 | 15 | 11 | 9 | 7 | 5 | |
| 1.4 | 340 | 165 | 107 | 78 | 60 | 48 | 40 | 34 | 29 | 25 | 18 | 13 | 10 | 8 | 6 | 4 | |
| 1.3 | 315 | 153 | 98 | 71 | 55 | 44 | 36 | 31 | 26 | 23 | 16 | 12 | 9 | 6 | 4 | 3 | |
| 1.2 | 290 | 140 | 90 | 65 | 50 | 40 | 33 | 28 | 23 | 20 | 14 | 10 | 7 | 5 | 3 | 2 | |
These depths are rounded down to the nearest metre.
See also
{{Portal bar|Underwater diving|Diving safety}}- {{annotated link|Oxygen toxicity}}
Notes
2. ^1 2 3 {{cite book |title=DAN Nitrox Workshop Proceedings |author=Lang, M.A. |year=2001 |publisher=Divers Alert Network |location=Durham, NC |url=http://archive.rubicon-foundation.org/4855 |accessdate=21 November 2012 |page=52}}
3. ^1 {{cite web |title = Physics of Diving |work = NOAA Diving Manual |publisher = National Oceanic and Atmospheric Administration |url = http://ehs.ucsb.edu/units/diving/dsp/forms/articles/physics.pdf |accessdate = 6 September 2013 |deadurl = yes |archiveurl = https://web.archive.org/web/20140531084518/http://ehs.ucsb.edu/units/diving/dsp/forms/articles/physics.pdf |archivedate = 31 May 2014 |df = }}
4. ^1 {{cite journal| last1 = Richardson| first1 = Drew| last2 = Shreeves| first2 = Karl| title = The PADI enriched air diver course and DSAT oxygen exposure limits| journal = South Pacific Underwater Medicine Society Journal| volume = 26| issue = 3| year = 1996| issn = 0813-1988| oclc = 16986801| url = http://archive.rubicon-foundation.org/6310| accessdate = 2 May 2008 }}
5. ^1 {{cite journal| last1 = Bitterman| first1 = N| last2 = Melamed| first2 = Y| last3 = Perlman| first3 = I| title = CNS oxygen toxicity in the rat: role of ambient illumination| journal = Undersea Biomedical Research| volume = 13| issue = 1| pages = 19–25| year = 1986| pmid = 3705247| url = http://archive.rubicon-foundation.org/3044| accessdate = 20 September 2008 }}
6. ^1 {{cite journal| last1 = Bitterman| first1 = N| last2 = Schaal| first2 = S| title = Caffeine attenuates CNS oxygen toxicity in rats| journal = Brain Research| volume = 696| issue = 1–2| pages = 250–3| year = 1995| pmid = 8574677| doi = 10.1016/0006-8993(95)00820-G }}
7. ^1 {{cite journal| editor1-last = Richardson| editor1-first = Drew| editor2-last = Menduno| editor2-first = Michael| editor3-last = Shreeves| editor3-first = Karl| title = Proceedings of rebreather forum 2.0| journal = Diving Science and Technology Workshop| year = 1996| pages = 286| url = http://archive.rubicon-foundation.org/7555| accessdate = 20 September 2008 }}
References
{{Reflist |refs=[1][2][3][4][5][6][7]}}
Sources
- {{cite book
| last1 = Clark
| first1 = James M
| last2 = Thom
| first2 = Stephen R
| year = 2003
| chapter = Oxygen under pressure
| pages = 358–418
| editor1-last = Brubakk
| editor1-first = Alf O
| editor2-last = Neuman
| editor2-first = Tom S
| title = Bennett and Elliott's physiology and medicine of diving
| edition = 5th
| publisher = Saunders
| location = United States
| isbn = 978-0-7020-2571-6
| oclc = 51607923
| ref = harv
- {{cite journal
| last = Donald
| first = Kenneth W
| year = 1947
| title = Oxygen Poisoning in Man: Part I
| journal = British Medical Journal
| volume = 1
| issue = 4506
| pages = 667–672
| pmc = 2053251
| pmid = 20248086
| doi = 10.1136/bmj.1.4506.667
| ref = {{sfnRef|Donald, Part I|1947}}
- {{cite book
| title = DAN nitrox workshop proceedings
| editor-last = Lang
| editor-first = Michael A
| year = 2001
| publisher = Divers Alert Network, 197 pages
| location = Durham, NC
| url = http://archive.rubicon-foundation.org/4855
| accessdate = 20 September 2008
| ref = harv
- {{cite book
|author = U.S. Navy Supervisor of Diving
|title = U.S. Navy Diving Manual
|version = SS521-AG-PRO-010 0910-LP-106-0957, revision 6 with Change A entered
|year = 2011
|publisher = U.S. Naval Sea Systems Command
|url = http://supsalv.org/pdf/Dive%20Manual%20Rev%206%20with%20Chg%20A.pdf
|format = PDF
|accessdate = 29 Jan 2015
|ref = {{sfnRef|U.S. Navy Diving Manual|2011}}
|deadurl = yes
|archiveurl = https://web.archive.org/web/20141210095431/http://www.supsalv.org/pdf/Dive%20Manual%20Rev%206%20with%20Chg%20A.pdf
|archivedate = 2014-12-10
|df =
}}{{Decompression}}{{Science of underwater diving}}
- Aliabad-e Kamfiruz
- Aliabad-e Kamfiruz-e Olya
- Aliabad-e Kamfiruz-e Sofla
- Aliabad-e Kankabud
- Aliabad-e Kapur Chal
- Aliabad-e Karaftu
- Aliabad-e Kareyak
- Aliabad-e Karvan
- Aliabad-e Kashantu
- Aliabad-e Kashmar
- Ali Abad -e- katool
- Aliabad-e Katool
- Aliabad-e Kavir
- Aliabad-e Kenar Shahr
- Aliabad-e Kerend
- Aliabad-e Khaleseh
- Aliabad-e Khan
- Aliabad-e Kharabeh
- Aliabad-e Khazayi
- Aliabad-e Khazayi-ye Aliabad-e Marki
- Aliabad-e Khenaman
- Aliabad-e Khosrowi
- Aliabad, Ekhtiarabad
- Aliabad-e Khvajeh
- Aliabad-e Khvoshablu