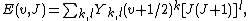- Relation to conventional band spectrum constants
- See also
- References
In quantum chemistry, the Dunham expansion is an expression for the rotational-vibrational energy levels of a diatomic molecule:
[1]where v and J are the vibrational and rotational quantum numbers.
The constant coefficients 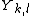 are called Dunham parameters with
are called Dunham parameters with 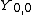 representing the electronic energy. The expression derives from a semiclassical treatment of a perturbational approach to deriving the energy levels.[2] The Dunham parameters are typically calculated by a least-squares fitting procedure of energy levels with the quantum numbers.
representing the electronic energy. The expression derives from a semiclassical treatment of a perturbational approach to deriving the energy levels.[2] The Dunham parameters are typically calculated by a least-squares fitting procedure of energy levels with the quantum numbers.
Relation to conventional band spectrum constants
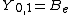 | 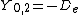 | 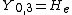 | 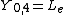 |
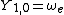 | 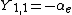 | 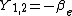 |
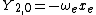 | 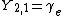 | |
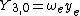 | ||
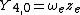 |
This table adapts the sign conventions from the book of Huber and Herzberg.
[3]See also
- Rotational-vibrational spectroscopy
References
1. ^{{cite journal| last=Dunham|first=J. L.|title=The Energy Levels of a Rotating Vibrator|journal=Phys. Rev. |year=1932|volume=41|pages=721–731|doi=10.1103/PhysRev.41.721|bibcode=1932PhRv...41..721D}}
2. ^{{cite journal|last=Inostroza|first=N. |author2=J.R. Letelier |author3=M.L. Senent|title=On the numerical determination of Dunham’s coefficients: An application to X1 R + HCl isotopomers|journal=Journal of Molecular Structure: THEOCHEM|year=2010|volume=947|pages=40–44|doi=10.1016/j.theochem.2010.01.037}}
3. ^{{cite book | title=Molecular Spectra and Molecular Structure IV. Constants of diatomic molecules | first1=K.P. | last1=Huber | first2=G. | last2=Herzberg | publisher=van Nostrand | year=1979 | location=New York | isbn=0-442-23394-9}}
{{applied-math-stub}}2. ^{{cite journal|last=Inostroza|first=N. |author2=J.R. Letelier |author3=M.L. Senent|title=On the numerical determination of Dunham’s coefficients: An application to X1 R + HCl isotopomers|journal=Journal of Molecular Structure: THEOCHEM|year=2010|volume=947|pages=40–44|doi=10.1016/j.theochem.2010.01.037}}
3. ^{{cite book | title=Molecular Spectra and Molecular Structure IV. Constants of diatomic molecules | first1=K.P. | last1=Huber | first2=G. | last2=Herzberg | publisher=van Nostrand | year=1979 | location=New York | isbn=0-442-23394-9}}
1 : Spectroscopy
- Estadio Sarría
- Estadio Sausalito
- Estadio Sergio Antonio Reyes
- Estadio Sergio León Chávez
- Estadio Sergio Torres
- Estadio Simeón Magaña
- Estadio Sixto Escobar
- Estadio Socios Fundadores
- Estadio Son Moix
- Estadio Sportivo Barracas
- Estadio Sánchez Pizjuán
- Estadio Tahuichi Aguilera
- Estadio TEC CCM
- Estadio TEC CEM
- Estadio Templo del Dolor
- Estadio templo del dolor
- Estadio Teodoro Lolo Fernández
- Estadio Teodoro Mariscal
- Estadio Tetelo Vargas
- Estadio Tiburcio Carías Andino
- Estadio Tomás Adolfo Ducó
- Estadio Tomás Oroz Gaytán
- Estadio tres de marzo
- Estadio Universidad del Mar
- Estadio universidad del mar